 
Ah, now it makes sense! When comparing two ferromagnets, one that is soft(narrow area), the other hard(wide area). So, these are the four processes in the ideal Brayton cycle. It's the loop part which needs clarity for the process of explanation and also practcal application. The difference between the work done by the gas and the work done on the gas is the network produced by the cycle and it corresponds to the area enclosed by the cycle curve (in Pv diagram below). 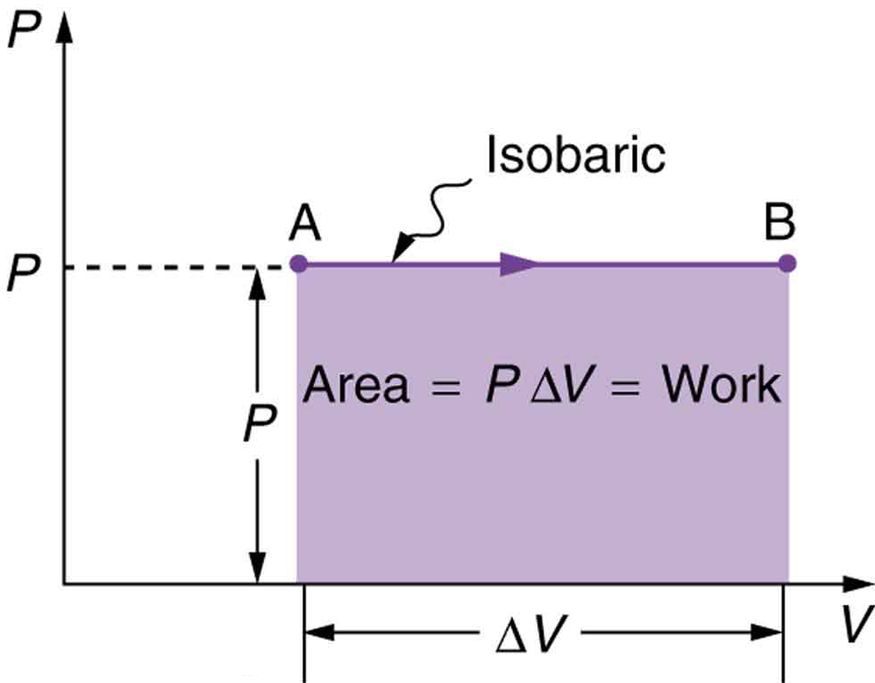
However, since the energy expended in doing this produces diminishing returns in terms of cost, the H field is limited to where saturation flattens the curve for most practical applications. To produce the maximum possible saturation remanance (m r) after the magnetising force is removed, requires a much larger H field than drawn on the curve. increasing the magnetising force beyond that point will not produce any further appreciable domain alignment. The curve flattens at the onset of saturation. H is the external magnetising field which forces alignment of the magnetic dipoles and crystaline domains within the ferromagnetic material. (On a linear scale, the hysteresis loop part of the curve would look extremely thin and saturation would look like a flat line with a very small gradient). 
It could be drawn on a log scale so that the whole curve could be accommodated, but serves no useful purpose to aid clarity. The H axis is limited to the portion of the curve where saturation starts. Is the energy dissipated defined as the work done by the system? I guess what has confused me is the definition of work done (although it's not excuse, since I should be remembering this now). The gauge enabled Watt to calculate the work done by the steam while ensuring that its pressure had dropped to zero by the end of the stroke, thereby ensuring that all useful energy had been extracted. The area between the curves in the loop is the difference in these two functions and represents the difference in the two amounts of energy and thus represents the energy lost in the cycle. But as the magnetic field B does not follow the same path in the reverse direction (demagnetisation) as the forward (magnetisation) there are two different mathematical functions here relating B and H. In that case the energy stored in the magnetic field from d to a would be recovered when the material was returned from a to d. The integral from a to d and the integral from d to a would indeed be equal but opposite in sign, if it was the same function (curve) in both directions. Force should be plotted on the vertical (y) axis Extension should be plotted on the horizontal (x). This, physically, represents work done here. A force-extension graph can be used to calculate the work done in joules when stretching a spring. We get the exact value of work done by making each interval so much small such that #916 x-> 0 which means curved path being decomposed into infinite number of line segment i.The area under a single curve is given by the integral of that function between the limits specified.Adiabatic Process (No Heat Transfer) An adiabatic system is perfectly insulated from external environment and there is no heat transfer in or out of the system. More accuracy of results can be obtained by making these interval infinitesimally smaller Work done in an isothermal process is given by, Thus for and expanding system work done is negative and for a shrinking system external work done is positive. Summation in equation 4 is equal to shaded area in figure 3(a).Total work done in moving the body from point A to point B.Work done in moving the body from x 1 to x 2 is given by.Now total displacement or path of the body can be decomposed into number of small intervals Δx such that with in each interval force F(x) can be considered to be approximately constant as shown below in the figure.For simplicity consider the direction of force acting on the body to be along x-axis also consider the force F(x) is some known function of position x.However, work can be done by forces that varies in magnitude and direction during the displacement of the body on which it acts.(a) In this change from state 1 to state 2, the volume increases and the work and area are. So far we have defined work done by a Force which is constant in both magnitude and direction. 6 The work done equals the area under the curve on a pV-diagram. From the first law of thermodynamics, the work done by turbine in an isentropic process can be calculated from: WT h3 h4s WTs cp (T3 T4s) From Ideal Gas Law we know, that the molar specific heat of a monatomic ideal gas is: Cv 3/2R 12.5 J/mol K and Cp Cv + R 5/2R 20. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
